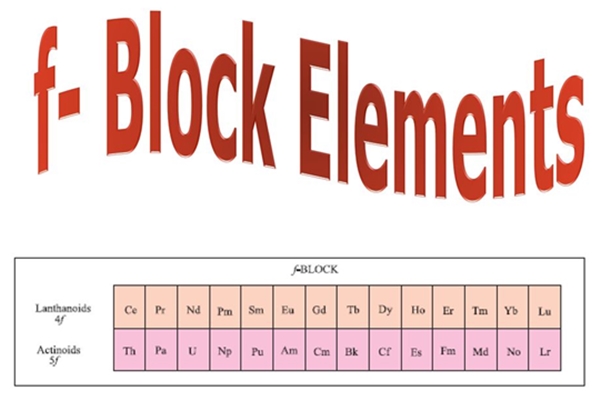
The f-block elements or inner transition elements are those in which the last electron enters any one out of seven f-orbitals of their respective ante-penultimate shells. These elements have electrons (1 to 14) in the f orbital, (0 to 1) in the penultimate energy level’s d orbital, and (0 to 1) in the outermost orbital. In the f-block, there are primarily two series that correspond to the filling of 4f and 5f orbitals. The elements are divided into two groups: the 4f series of Ce to Lu and the 5f series of Th to Lw. Each series contains 14 elements that fill the ‘f’ orbital.
Classification
Inner transition elements are typically members of Group 3, but they can also be viewed separately as f block elements. The 4f and 5f orbitals are filled in the f-block elements. The f-block elements are the inner transition elements. These metal elements provide a transition in the sixth and seventh rows of the periodic table, separating the s block and the d block elements.
The F-block elements are classified into two series- lanthanides and actinoids.
- The first series of the f block elements includes elements with atomic numbers ranging from 57 to 71. These substances are not radioactive (except for promethium). The last electron in the lanthanide series enters the 4f orbital. This is called the lanthanide series.
- The actinide series consists of elements with atomic numbers ranging from 89 to 103. In nature, these elements are generally radioactive. The final electron in the actinide series enters the 5f orbital.
Properties of Lanthanides
Lanthanide or lanthanoid chemical elements are a group of 15 metallic chemical elements with atomic numbers 57-71, ranging from lanthanum to lutetium.
- The atomic radius decreases as the lanthanide series progresses. This decrease is due to lanthanoid contraction. Lanthanoid contraction occurs as a result of the 4f orbital’s poor shielding. The outermost electron is affected by the positive nuclear charge as a result of this inadequate shielding. As a result, the atomic radius decreases with the lanthanide series.
- Lanthanoids typically have +3 oxidation states, but they can also have +4 and +2 oxidation states. This variation in oxidation states is caused by the f-orbital being fully filled, partially filled, or empty.
- Metals make up the majority of the elements in the lanthanide series. They are excellent electrical and thermal conductors. The hardness of the metal increases as the atomic number increases.
Properties of Actinides
The actinide or actinoid series includes the 15 metallic chemical elements actinium through lawrencium with atomic numbers ranging from 89 to 103. The name actinide comes from the first element in the series, actinium.
The atomic radius decreases with the series as a result of actinoid contraction. This contraction occurs as a result of the 5f orbitals‘ poor shielding. As a result, the outermost electron is affected by the positive nuclear charge, which reduces the atomic radius and the actinide series.
The most common oxidation state shown by actinoids is +3 (but this is not always stable). The metals in the actinides series are highly reactive.
Summary
- There are 30 elements belonging to the f-block.
- The last electron of all the elements enters the f-orbital.
- The f-block is also known as the Inner transition elements.
- The elements from 57 – 71 are lanthanides.
- The elements from 89 – 103 are actinides.
- All the elements of the f-block are metals.